Photocatalytic degradations of organic pollutants in wastewater using hydrothermally grown ZnO nanoparticles
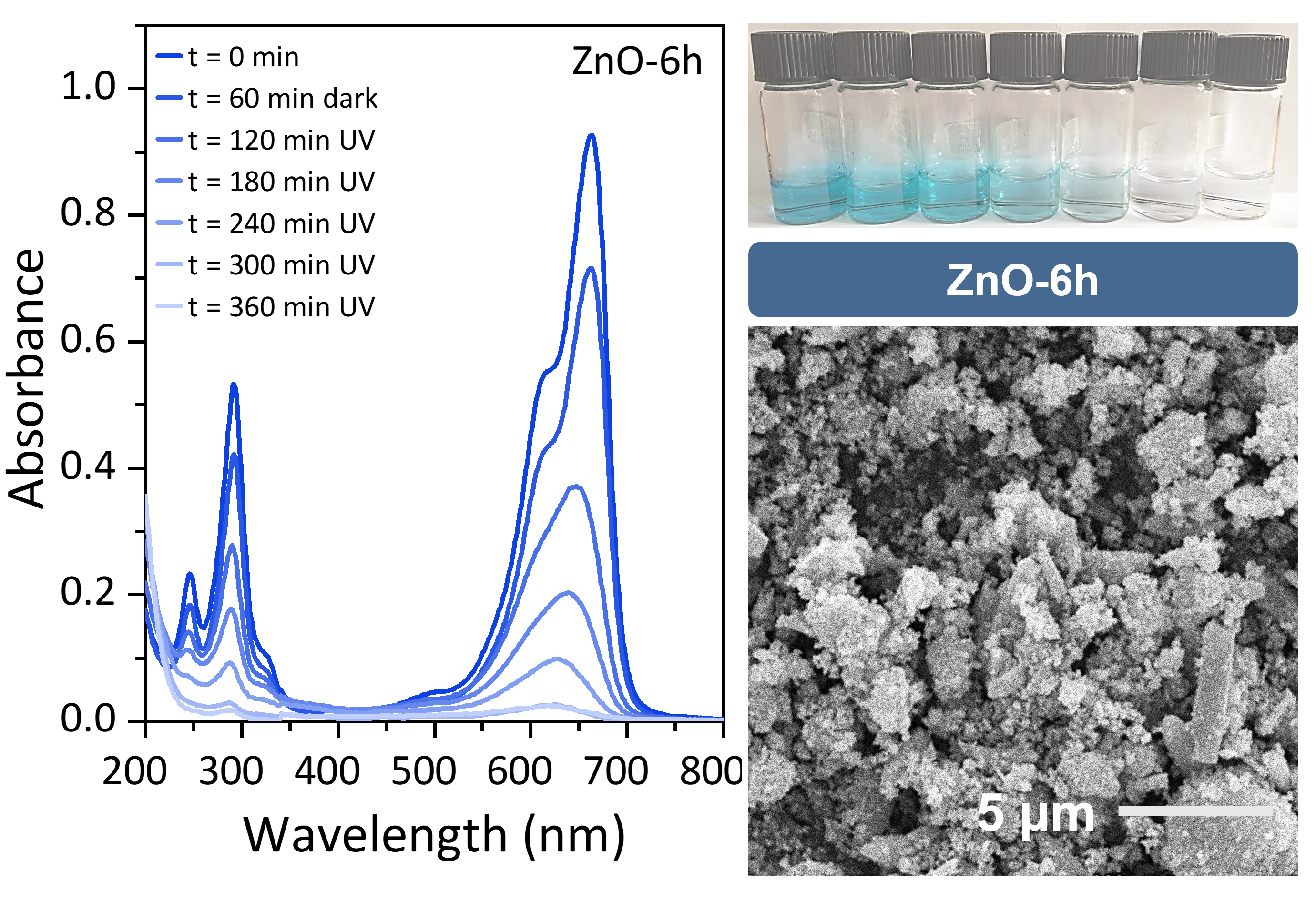
The increasing prevalence of organic pollutants in wastewater poses a significant environmental challenge due to their persistence and harmful effects. Photocatalysis using semiconductor nanoparticles, such as ZnO, has emerged as a promising approach for pollutant degradation, but optimizing the structural and functional properties of these materials remains a critical challenge. In this study, ZnO nanoparticles were synthesized via a hydrothermal method with varying durations (4, 6, and 8 hours) to investigate the impact of synthesis time on their photocatalytic efficiency. The structural and compositional properties were characterized using SEM, XRD, and EDS analyses, revealing that longer synthesis times improve crystallinity and alter the Zn:O atomic ratio, affecting defect density and stoichiometry. Photocatalytic performance was evaluated through the degradation of an organic pollutant under UV illumination. ZnO-6h exhibited the highest rate constant (k=0.017 min−1), outperforming ZnO-4h (k=0.016 min−1) and ZnO-8h (k=0.013 min−1). This superior activity is attributed to an optimal combination of high crystallinity, intermediate morphology, and the presence of oxygen vacancies that enhance charge carrier dynamics. The findings demonstrate that synthesis duration is a critical parameter in tuning the structural and photocatalytic properties of ZnO nanoparticles. This study provides insights into the design of ZnO-based photocatalysts and underscores their potential for environmental remediation. Future research could extend these findings by exploring scalability and pollutant-specific applications, paving the way for more efficient wastewater treatment technologies.